SPAI
Home » News
EMA report on veterinary drugs published
The 2022 Annual Report for regulatory activities in the veterinary medicines sector has been published by the European Medicines Agency (EMA). Ten new veterinary medicines were authorised in Europe in 2022: these included two vaccines, and three treatments with new active ingredients.[…]
Read MoreMedical devices: what happens when nonconformities relating to stages of production effected by suppliers are detected?
When a medical device production process is defined, some of the production tasks may be outsourced to third party suppliers for technological and/or organisational reasons. Of course, this requires regular checks on the produced items, the manufacturing processes and the materials used[…]
Read MoreThe pharmaceutical industry is increasingly important to the Italian economy
According to the Nomisma report on the so-called “Fab13” of the pharmaceutical sector (the 13 Italian-capital sample companies), global growth in the sector saw a 12.6% increase in revenues compared to 2021 with a 14.9% increase in exports (72% of total sales).[…]
Read MoreWill manufacturers of sterile medicines be able to implement GMP Annex 1 requirements by August?
In August 2022, the European Commission published the revised edition of Annex 1 “Manufacture of sterile medicinal products” in EudraLex – Volume 4 – Good Manufacturing Practices (GMP) guidelines. This document will have a significant impact on manufacturers of sterile medicines who[…]
Read MoreVeterinary medicines: EU Regulation 2022/1644 in force
Commission Delegated Regulation 2022/1644 which governs official controls on the use of pharmacologically active substances in DPA animals (intended for food production) came into force on 15 December 2022. It follows that the transitional measures which had, up until 14 December 2022,[…]
Read MorePharma 4.0 and cybersecurity
The Cost of a Data Breach Report 2020 by IBM and the Ponemon Institute showed that pharmaceutical and biotech companies suffer the highest number of cyber attacks (or ‘breaches’). The reason for this lies in the fact that the pharmaceutical sector is[…]
Read MoreSecond edition of GAMP 5 published
In July the ISPE (International Society for Pharmaceutical Engineering) published the second edition of the GAMP 5 guidelines for validating IT systems in the pharmaceutical sector. GAMP 5 guidelines are now a standard used by the regulatory bodies themselves during inspections. First[…]
Read MoreNew revision for Annex 1
Fourteen years in the making and two draft versions submitted in 2017 and 2020, on 25 August 2022 the final version of the new Annex 1 of the GMP (Good Manufacturing Practice) was published on the EudraLex website. Receiving over 6,000 comments[…]
Read MoreThe most beautiful silence you’ve ever listened to
A few weeks ago, a world leader in the pharmaceutical packaging sector issued the official version of a digital product, namely a self-explanatory course intended for the foreign purchasers of a Primary packaging line. I was the appointed project manager of this digital product for technical training[…]
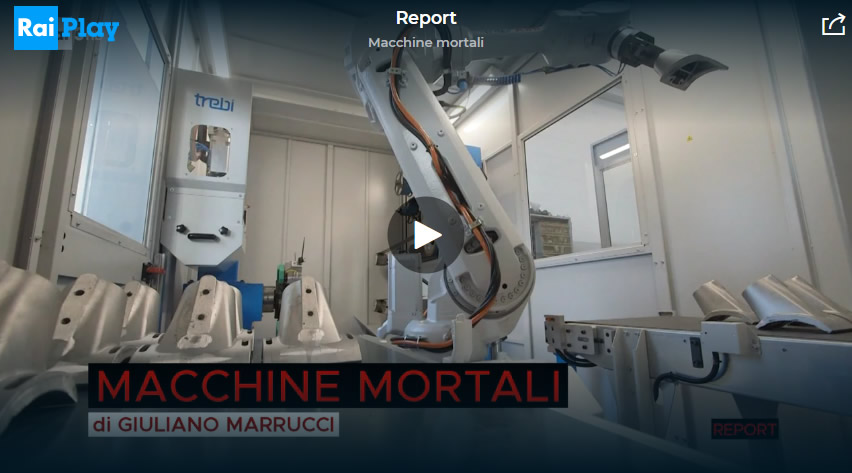
Read MoreMachines and workplace accidents
Deaths in the workplace were the subject of an episode of the Italian TV programme Report, which aired on channel Rai 3 on 9 May. While the European average is 2.2 deaths per 100,000 employees, Italy stands at 2.7, which is much[…]
Read More